CCC’s Monthly Digest serves to inform its members of developments from the previous month concerning topics of interest to the Council. Below are highlights which cover past, current, and on-going regulatory updates, industry & consumer issues, and Council activities from June 2018.
US Policy and Regulatory Updates
• FDA and USDA Cooperate to Streamline Efforts
• Trump Proposes Consolidating Food Safety Under Single Agency Within USDA
• Industry Coalition Pushes for Amendment to Fair Packaging and Labeling Act
• FDA Reconsiders Approach to Declaration of Added Sugars on Pure Maple Syrup and Honey
• FDA Releases the First of Three Draft Guidances on FSMA Food Defense Requirements
• California Judge Rules that Glyphosate Does Not Need Cancer Warning
International Policy and Regulatory Updates
• WHO Leaves Out Endorsement for Soda Taxes
• Australian Beverage Industry Refuses to Reformulate
• India’s Proposed New Food Safety and Standards (Labeling and Display) Regulations Meet Criticism
• Translation of Chinese Standard for Maximum Levels of Contaminants in Foods
Publication Updates
• IFIC Survey Indicates Shift in Consumer Attitudes toward Sugar & Carbohydrates
• Preliminary Study Results Indicate U.S. Obesity Rates Rising Again
• GMA’s SmartLabel Marks Sevenfold Growth as Consumers Seek Transparency
• Non-GMO Project Predicted to Profit from Federal BE Legislation
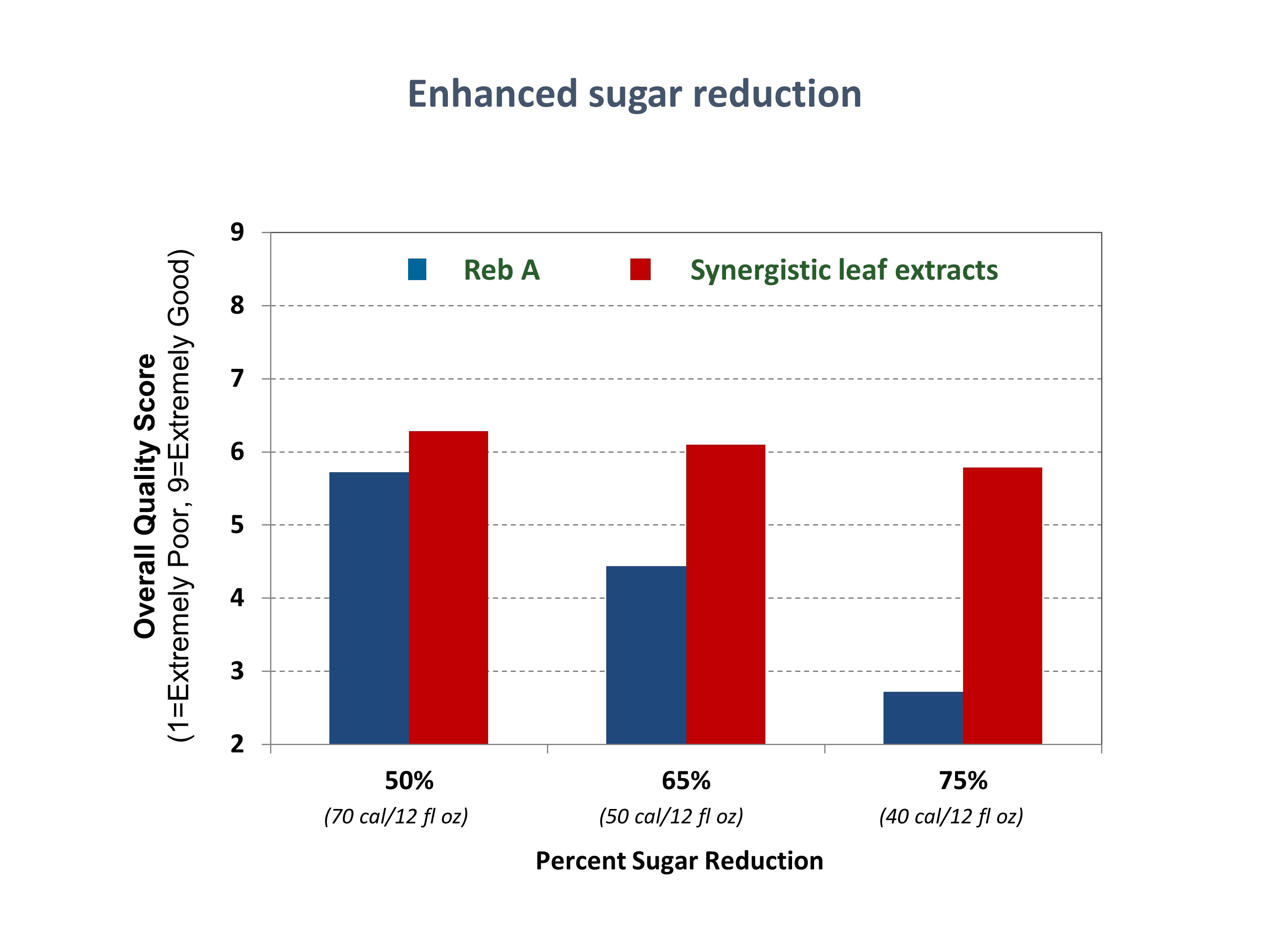
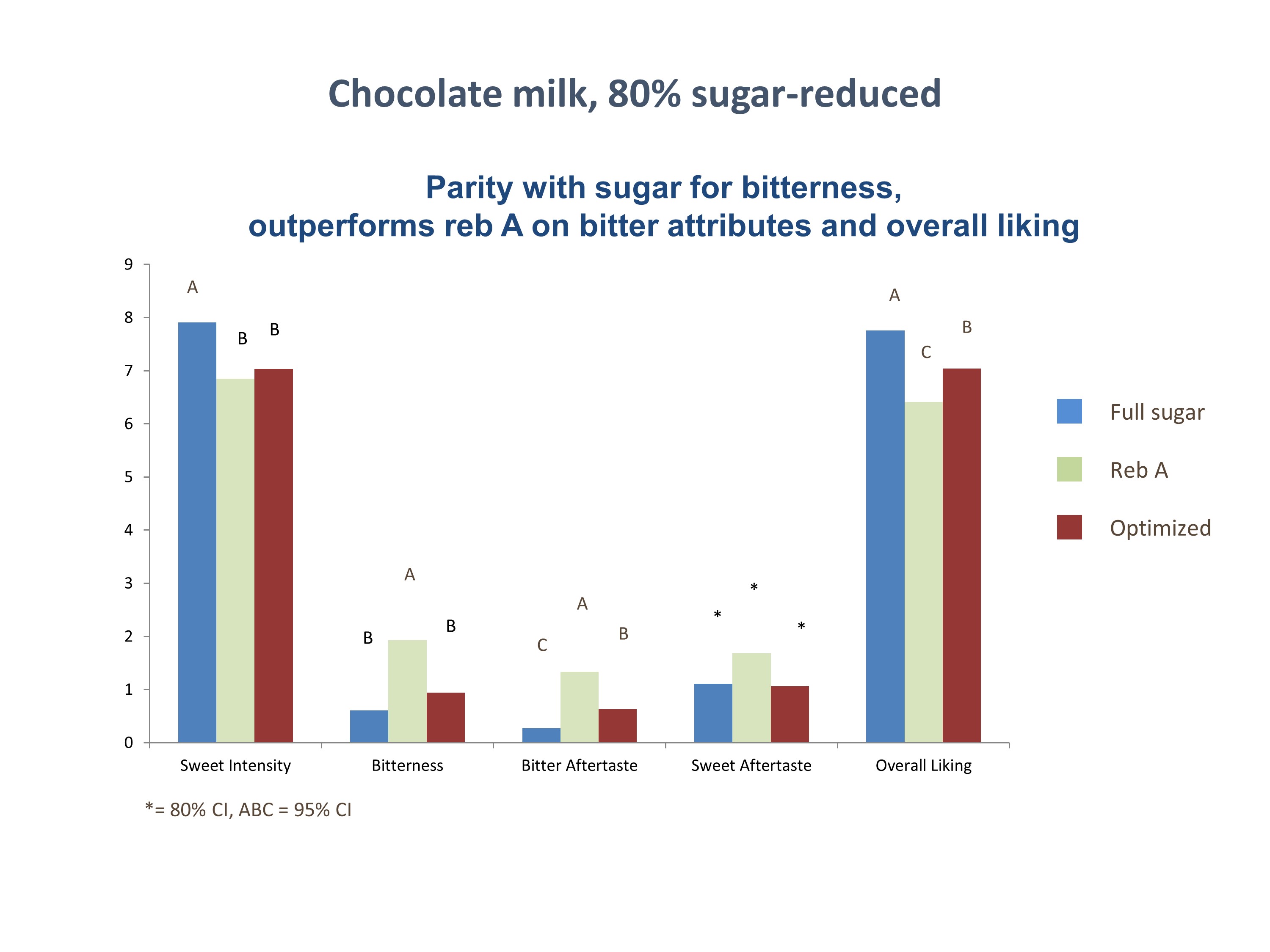
• Reformulation Experts Weigh in on Sugar Reduction at Summit in Singapore
• CDC Study Shows Rural America has Higher Obesity Rates than Urban Areas
• EU Consumer Organization Publishes Report on Food Labeling
• Survey Says Graphics More Effective Than Text in Sugar Warning Labels
• 2018 Sweetener Systems Conference to Focus on Sweeteners and Sugar Reduction
Council Updates
• CCC Staff Attends Nutrition 2018 – Annual Conference by the American Society for Nutrition
• CCC Submits Comments to FSSAI
• Communications Update
US Policy and Regulatory Updates
FDA and USDA Cooperate to Streamline Efforts
On May 30, the U.S. Department of Agriculture (USDA) announced that Secretary Sonny Perdue and the U.S. Food and Drug Administration’s (FDA) Commissioner Scott Gottlieb signed an agreement formalizing efforts to coordinate research in food safety and related regulatory issues. The agreement seeks to increase clarity, efficiency, and potentially reduce the number of duplicate regulations subject to oversight by both agencies. USDA notes this streamlining can reduce costs on industry and free up government resources to target areas of high risk.
In addition, the statement also reports on efforts by the two agencies to collaborate on scientific research. It is noted that USDA’s Office of the Chief Scientist (OCS) recently hosted a seminar for USDA scientists to hear from FDA scientists on the modernizations and exposure assessments of the Total Diet Study (TDS), an ongoing FDA program that monitors levels of about 800 contaminants and nutrients in the average U.S. diet.
Trump Proposes Consolidating Food Safety Under Single Agency Within USDA
On June 22, Food Safety News reported President Trump wants to consolidate the USDA’s Food Safety and Inspection Services (FSIS) and the food safety functions of FDA into a single agency housed in the USDA called the Federal Food Safety Agency. The article notes former Presidents’ similar failed attempts to consolidate food safety efforts under the U.S. Department of Health and Human Services (HHS). While Congress did not extend the power to President Obama to reorganize the government, President Trump is seeking executive reorganization authority under his “Delivering Government Solutions in the 21st Century” report released on June 21, 2018. The plan also recommends several other changes for USDA and HHS, including consolidating non-commodity nutrition assistance programs and moving them from USDA to HHS, and renaming HHS the Department of Health and Public Welfare.
The new USDA food safety unit would bring on about 14,200 employees with an annual budget authority of about $2.3 billion by combining existing FSIS and FDA food safety workforces and budgets. If the Trump reorganization were to go through, FDA would be renamed the “Federal Drug Administration” and continue with its focus on drugs, medical devices, biologics, tobacco, cosmetics, and dietary supplements, which would still be in the category of food.
As reported by Food Chemical News (subscription required), while there is both support for and opposition to the proposed consolidation, the overwhelming response is doubt it will actually happen because it would require approval from Congress, which experts say is highly unlikely. Brian Ronholm, former deputy undersecretary for food safety at FSIS during the Obama administration, suggested that if the merger were to happen, industry may have more leverage at USDA than it would within a public health agency. However, Ronholm notes that the different cultures and approaches could be complicated to combine, and stated “I think it’s fair to say that FDA is publicly viewed as a having a more science-based culture, while the culture at FSIS is focused primarily on inspections, particularly the day-to-day inspections,” and, “the food safety functions at USDA are still very old, and they need modernization. With FDA, the nature and frequency of inspection is widely different […] and that’s just another issue to consider when you’re undertaking something like this.” An article published by The Conversation on June 24 outlines specific reasons why Trump’s plan to consolidate federal food safety efforts won’t work, and concludes by stating that the Trump administration may have to settle for the less ambitious goal of better interagency coordination to address concerns about duplication between the agencies’ scopes of work.
Industry Coalition Pushes for Amendment to Fair Packaging and Labeling Act
As reported by Food Navigator in a June 13 article, the Coalition for Accurate Product Labels (CAPL), comprised of over 60 organizations including the Grocery Manufacturers Association, the Independent Bakers Association, and the National Potato Council, have joined together to ask Congress to amend the Fair Packaging and Labeling Act. The coalition is calling on Congress to pass a bill which would establish a federal standard for risk-based labeling and ingredient disclosure requirements in an effort to preempt state initiatives from requiring warning labels that go beyond national standards, such as proposals in New York, San Francisco, and Baltimore for warning labels on sweetened beverages. According to the coalition’s proposed amendments, states would have to meet science-based criteria before enacting their own ingredient listing or labeling programs. In addition, the group is pushing for permission to allow state-mandated product information to be provided through smartphone-enabled “smart labels” and online. Finally, the coalition is requesting that all covered product information be risk-based, noting “Warning labels act as important precautionary notices of legitimate risks and need to be taken seriously.”
CCC has not joined CAPL based on previous member feedback. If you support CCC revisiting this decision, please advise Council staff.
FDA Reconsiders Approach to Declaration of Added Sugars on Pure Maple Syrup and Honey
On June 19, FDA announced in a press release that after receiving more than 3,000 comments on its draft guidance for declaring added sugars on honey, maple syrup, and certain cranberry products, the agency recognizes the complexity of the issue and plans to “swiftly formulate a revised approach that makes key information available to consumers in a workable way.”
The draft guidance was released in February 2018 and was proposed to help industry provide information about the added sugars declaration on the label of products like maple syrup and honey, which are defined as packaged “added sugars” in FDA’s final rule based on the 2015-2020 Dietary Guidelines for Americans. In an effort to address concerns that declaring added sugars on the Nutrition Facts label of single ingredient products like pure maple syrup and pure honey may lead consumers to think that a “pure” product is economically adulterated with corn syrup or other added sweeteners, the guidance noted FDA’s intent to exercise enforcement discretion to allow the use of an obelisk symbol on the label after the added sugars percent daily value on containers of pure maple syrup or pure honey which would direct consumers to language outside the label that provides non-misleading information about the meaning of “added sugars” for these specific products. The overwhelming feedback received by FDA is that the approach laid out in the draft guidance does not provide the clarity that the agency intended.
FDA Releases the First of Three Draft Guidances on FSMA Food Defense Requirements
On June 20, FDA announced in the Federal Register the release of the first four chapters of a nine-chapter draft guidance on protections against intentional adulteration (the “IA rule”). As noted in a Covington Alert, these first four chapters focus on the components of the food defense plan, including how to conduct vulnerability assessments and how to identify and implement mitigation strategies and food defense monitoring requirements.
FDA intends to release the remaining chapters of the IA draft guidance in two other installments “later this year.” The second group of chapters will focus on a flexible vulnerability assessment approach. The third set of chapters will provide greater detail on how to take corrective action, how to verify that a facility’s system is working, food defense plan re-analysis requirements, and record-keeping requirements.
California Judge Rules that Glyphosate Does Not Need Cancer Warning
As reported by AgWeb on June 13, a California judge ruled on June 12 that glyphosate does not need a label warning it causes cancer. The judge stated, “It is inherently misleading for a warning to state that a chemical is known to the state of California to cause cancer based on the finding of only one organization – International Agency for Research on Cancer (IARC), when apparently all other regulatory and governmental bodies have found the opposite, including EPA.”
Although glyphosate is not a substance of interest to CCC, the decision to uphold the California Federal District Court’s partial injunction against the Office of Environmental Health Hazard Assessment (OEHHA) based on the premise that the glyphosate warning is “inherently misleading” is seen by many as precedent-setting. As CCC members look towards a potential IARC review of specific sweeteners, and recognizing OEHHA’s Chemical Identification Committee considered aspartame for possible prioritization and listing on Proposition 65 in 2016, this action indicates that a robust body of safety evidence, and support will likely supersede the outcome of an IARC review.
International Policy and Regulatory Updates
WHO Leaves Out Endorsement for Soda Taxes
As discussed in a June 5 article in Food Chemical News (subscription required), the World Health Organization (WHO) published a report on June 1 recommending ways to prevent early death and non-communicable diseases (NCD) such as diabetes and heart disease. While the report makes a variety of recommendations to promote healthier eating, it notably leaves out an endorsement for soda taxes, going against the WHO’s strong previous position in favor of taxing sugary drinks to help reduce sugar intake. The article notes that the 26 members of the Commission were swayed to avoid including this recommendation in the current report by Deputy Secretary Hargan, whose spokesperson later confirmed in a statement, “The terms of reference for the Commission said recommendations should be bold to enable countries to accelerate progress on NCDs. Taxes on sugary drinks is not new, bold, or innovative. More importantly, evidence is lacking that such a tax produces positive health outcomes, specifically reducing the burden of NCDs.”
While soda taxes are not addressed, the report suggests governments should work with food and non-alcoholic beverage companies in areas such as reformulation, labeling, and regulating marketing.The International Food and Beverage Alliance (IFBA) has voiced support for the report’s recommendations directed at empowering consumers to “eat balanced diets and live healthier lives.”
Australian Beverage Industry Refuses to Reformulate
According to a June 12 article in FoodNavigator, the Australian Beverage Council and one of the largest bottlers of ready-to-drink beverages in the Asia-Pacific region, have refuted rumors that Australia plans to further lower the amount of sugar in their products, specifically reaching a 20% sugar reduction by 2020, to avoid a potential sugar tax. While there are currently 28 countries that have introduced taxes on sugar-sweetened beverages (SSBs), Australia’s government reported that there will not be a tax on SSBs after the next elections in 2019. The major bottling company stated that their goal remains at the overall reduction of 10% across all products sold by 2020.
India’s Proposed New Food Safety and Standards (Labeling and Display) Regulations Meet Criticism
According to a June 20 article in FoodNavigator, the Center for Science and Environment (CSE), a public interest research and advocacy organization, submitted a list of recommendations on draft regulations released by the Food Safety and Standards Authority of India (FSSAI) in April, which include new mandatory front-of-pack labelling for packaged food products, including a red light label for foods high in fat, sugar and/or salt. While CSE has expressed its support for these regulations, the organization highlighted the following gaps:
- The draft does not provide specific requirements for the labeling of added sugar and dietary fiber.
- The draft simply states that foods high in fat, sugar, and salt should not be advertised to children in any form. CSE believes this is insufficient.
- Although the draft emphasizes providing nutrition information for each serving of a food item and states that the percentage of salt, sugar, and fat contributing to the total daily recommendation for each should be communicated, the draft does not standardize serving sizes. Instead, the determination of serving sizes has been left to industry.
- CSE believes the standard set regarding genetically modified (GM) food is very weak, and recommends that FSSAI regulate “illegal GM food in India” while also setting a stricter bar for receiving exemption from GM labelling.
The article reports that FSSAI plans to finalize these regulations within the next two to three months. As a reminder, CCC submitted comments on FSSAI’s draft regulations due June 10 (see Council update below).
Translation of Chinese Standard for Maximum Levels of Contaminants in Foods
USDA’s Global Agricultural Information Network (GAIN) published a report on June 21 which included a translation of the China National Health and Family Planning Commission (NHFPC) and China Food and Drug Administration (CFDA)’s National Food Safety Standard for Maximum Levels of Contaminants in Foods. The standard, which was originally released in March 2017 and went into effect September, sets limits for lead, cadmium, mercury, arsenic, tin, nickel, chromium, nitrite, nitrate, Benzo[a]pyrene, N-nitrosodimethylamine and polychlorinated biphenyl, 3- chloro-1, 2-propanediol in foods.
Publication Updates
IFIC Survey Indicates Shift in Consumer Attitudes toward Sugar & Carbohydrates
A June 8 article in FoodNavigator reported on the recently-published International Food Information Council’s (IFIC) annual food & health survey, an online survey of approximately 1,000 Americans ages 18 to 80. The survey found that a significantly higher percentage of consumers believe that sugars are the source of calories most likely to cause weight gain. Specific survey findings of interest to the Council are included below:
- 33% of Americans believe sugars are the source of calories most likely to cause weight gain compared with 11% in 2011.
- 77% said they are trying to limit or avoid sugars. Of those, 60% are drinking more water instead of caloric beverages, and 20% said they are switching to low or no calorie beverage options.
- 45% had a negative attitude towards low or zero calorie sweeteners, although the survey did not differentiate between specific sweeteners.
- When asked to compare two products with an identical Nutrition Facts panel but different claims on pack, those with non-GMO claims, shorter ingredients lists, a less sweet taste, or ‘green’ credentials were perceived to be healthier.
Preliminary Study Results Indicate U.S. Obesity Rates Rising Again
An article published on June 12 by Health Day News covered a preliminary study led by Dr. Youfa Wang, a professor at Ball State University, presented during the 2018 American Society for Nutrition (ASN) annual meeting June 9-12 which was attended by CCC staff. The study’s findings show the U.S. obesity rate may be climbing again, after briefly leveling off in recent years. It is reported that, if nothing changes, half of all U.S. teenagers will be overweight or obese by 2030, as will one-third of children ages 6 to 11. However, when it comes to broader efforts, Wang said studies have shown some bright spots. For example, “moderate” evidence that school programs focused on diet and exercise can be helpful.
GMA’s SmartLabel Marks Sevenfold Growth as Consumers Seek Transparency
On June 12, Supermarket News reported that consumers’ desire to know more about the foods they purchase has caused a sevenfold increase in products using SmartLabel since early last year. SmartLabel is a digital tool accessed by scanning a product’s QR code which then links consumers to information on why ingredients are in the product, what they do, and how they are made, in addition to allergen descriptions, usage instructions, and sustainability.
The article notes key findings from a study sponsored by the Grocery Manufacturers Association (GMA) and the Food Marketing Institute (FMI). According to the survey of approximately 1,000 U.S. adults classified as primary shoppers, more than 70 percent said that they want more product information than what is on traditional package labels. In addition, 80 percent of grocery shoppers polled said they’re more likely to buy an item if its ingredient list is easy to understand. Further, 75 percent indicated they would alter their grocery buying habits if they had more information on a product, such as its environmental impact, safety and usage.
Non-GMO Project Predicted to Profit from Federal BE Legislation
A June 13 article in FoodNavigator covered the Non-GMO Project’s (NGP) responses to the USDA’s Proposed Rule establishing a National Bioengineered Disclosure Standard which was published in the Federal Register on May 4. The article includes a comment by the Non-GMO Project’s executive director Megan Westgate who stated that the NGP will “only become more relevant” as the mandatory bioengineered labeling law is implemented, partly because “…its scope could be so narrow that consumers will not simply be able to assume that anything without a bioengineered label is automatically non-GMO.” Westgate claims that given that thousands of products that have been produced with genetic engineering at some stage of manufacturing, they may not be subject to the mandatory GMO labeling under the proposed rule, consumers that really care about the issue will look for the Non-GMO Project Verified symbol when they shop.
The NGP argues that the definition of bioengineered is too narrow, and is advocating that the USDA use the Codex definition of biotechnology so as to mitigate trade disputes. It is also noted that many newer genetic engineering techniques are not covered in USDA’s definition, including CRISPR and RNAi, and there appears to be misalignment on this issue, as USDA includes apples and potatoes that have been developed using RNAi techniques as examples of not highly adopted crops.
Like many other industry stakeholders, the NGP feels that the proposed rule contains more questions than answers. However, the NGP strongly objects to the use of the term “bioengineered” and the accompanying logo, arguing that “Bioengineering really is a medical term. It’s not a term that’s used in the food space or by the public. They [USDA] should use terms that the public recognizes or it is fundamentally misleading …”
CCC is considering commenting on the proposed rule, as well as co-signing comments from larger industry coalitions such as the Coalition for Safe and Affordable Food and the Food & Beverage Issue Alliance.
Reformulation Experts Weigh in on Sugar Reduction at Summit in Singapore
As reported in a June 14 article on FoodNavigator, reformulation experts are warning food and beverage manufacturers against seeking to create “wonder foods” to solve growing health problems in the Asia Pacific region. During the summit, Dr. Kalpana Bhaskaran presented on the complications and barriers involved in trying to respond to consumer demand for reformulated foods such as “ice cream with increased fiber, functional ingredients and a memory booster, while also being low in sugar and containing all natural ingredients.” Kalpana explained key considerations that must be addressed by companies looking to reformulate, including technical incompatibility when combining different ingredients and the availability, capabilities, and cost of production facilities and resources.
CDC Study Shows Rural America has Higher Obesity Rates than Urban Areas
On June 15, the U.S. Centers for Disease Control and Prevention (CDC) released an article entitled “Obesity Prevalence Among Adults Living in Metropolitan and Nonmetropolitan Counties – United States, 2016.” The report identifies differences in obesity prevalence by metropolitan status, and found that obesity prevalence was significantly higher among adults living in nonmetropolitan (rural) counties (34%) than among those living in metropolitan (urban) counties (29%). The greatest differences in obesity prevalence was found between rural and urban residents in the South and Northeast regions.Findings in this article are consistent with those previously reported using 2005-2008 NHANES data. While CCC does not target specific geographic areas within the U.S., staff continues to monitor for opportunities to reach out to health care professional (HCPs) working to improve outcomes of those affected by diet-related diseases.
EU Consumer Organization Publishes Report on Food Labeling
Earlier this month, BEUC, known as the European Consumer Organization comprised of 43 independent national consumer groups from 32 European countries, published a report entitled “Food Labels: Tricks of the Trade – Our recipe for honest labels in the EU.” The 72-page report is based on BEUC’s alleged finding that there is a high prevalence of “food labeling tricks” used by food companies across Europe who are taking advantage of grey areas within EU food labeling laws. The report separates the types of misleading labeling techniques into three categories: quality, fruits, and whole grain. Regarding quality, BEUC requests that the EU legally define or at least harmonize guidance for common quality descriptions such as ‘traditional’ and ‘natural.’ Regarding fruit, BEUC demands that manufacturers should not be able to use any pictures of fruit on front packaging if there is no actual fruit content, and that, if fruit is used, the percentage should be shown clearly on the front pack. Further, minimum fruit content levels should be set by the EU for products who wish to use graphics, words or pictures to suggest fruit content on their label. In the report’s conclusion, BEUC recommends that the percentage of all advertised ingredients should be clearly displayed on labels.
Survey Says Graphics More Effective Than Text in Sugar Warning Labels
As reported by a June 19 article in Health Day, a study conducted by researchers at the Harvard School of Public Health entitled “The Effect of Graphic Warnings on Sugary-Drink Purchasing” was published on June 18 in the journal Psychological Science. The study is comprised of a primary field study and two follow-up studies online.
Researchers tested three different types of labels addressing sugar sweetened beverages: text warnings about health risks, graphic warnings about health risks, and listings of the caloric value of the drinks. The three labels were tested consecutively by placing each near bottled and fountain beverages in a hospital cafeteria in Massachusetts. In this initial study, researchers found that the portion of sugar-sweetened beverages purchased in the cafeteria declined by 14.8% when graphic warnings were displayed, whereas purchases of water increased. However, text warnings and calorie labels did not have a significant effect on purchasing behavior.
The remaining two follow-up studies were conducted online. The first, which asked consumers how the graphic warning label would influence their drink purchases, found that the graphic warnings prompted increased consideration of health risks over taste. The second, asked whether participants would support putting the three labels on sugar-sweetened beverages and found equal support for graphic labels, text warnings, and calorie labels.
2018 Sweetener Systems Conference to Focus on Sweeteners and Sugar Reduction
Global Food Forums is holding its 3rd annual Sweetener Systems Conference on October 23, 2018 in Oak Brook, Illinois. This year’s program will cover properties and interactions that impact the sweetness perception and technical function of finished products. Sessions will include updates on consumer attitudes, reformulating for sugar reduction, critical regulatory updates, emerging technologies, and nutritional implications.Side effects of alternative sweeteners including off-tastes, delayed sweetness onset, adaptation/desensitization effects and mouthfeel will be covered. Registration is offered at a discounted rate until August 31.
Council Updates
CCC Staff Attends Nutrition 2018 – Annual Conference by the American Society for Nutrition
CCC staff was able to attend a number of highly relevant sessions during the American Society for Nutrition Annual Conference in Boston, MA, June 9-12. While there were a number of sessions relevant to low- and no- calories sweeteners and carbohydrates, there were two that highlighted the need for future research on sweeteners and health including recommendations on how stakeholders should work together to conduct the research that is actually needed. These sessions included:
“Conflicting Outcomes from Systematic Reviews: Is the Consumption of Low Calorie Sweeteners a Benefit or a Risk for Weight Management?” and “Sweetness, Sweeteners and Health.”
CCC Submits Comments to FSSAI
On June 10, CCC submitted the attached comments to the Food Safety and Standards Authority of India (FSSAI) notice calling for public comments on the draft Food Safety and Standards (Labelling and Display) Regulations, 2018 (F.No 1-94/FSSAI/SP(Labelling)/2014(Pt-2)). CCC’s comments included opposition to proposed declarations on foods containing sweeteners, declarations on advertisements for products containing sweeteners, and a statement, “not recommended for children on products with sweeteners and advertisements for products that include sweeteners.”
Communications Update
- American Diabetes Association’s 78th Scientific Sessions: In response to two possibly negative presentations covering non-nutritive sweeteners presented at the conference, staff ran an add promoting the benefits of low- and no-calorie sweeteners in the Monday, June 25th addition of DiabetesPro SmartBrief.
- World Health Organization (WHO) Nutrition Guidance Expert Advisory Group (NUGAG): In advance of pending news affecting LNCS, staff reached out to 30 key media contacts in preparation of the release of NUGAG’s guidelines on carbohydrates and non-sugar sweeteners, to encourage them to use CCC as a resource.
- Working Group
- Allulose: Staff created and launched advertising targeted to food scientists, reaching them through the Institute for Food Technologists (IFT) via its website, magazine and e-newsletter.
- Stevia: In collaboration with the International Stevia Council, staff developed and began promotion of Dr. John Fry’s Stevia research “Sensory Analysis and Mathematical Modelling Drive Improved Stevia Taste” to food scientists, reaching them through IFT, also via the website and e-newsletter.